Study Shows Use of Obesity Drugs Alongside Behavioral Intervention is Associated with Reduced “Food Noise”
May 12, 2026 · Baton Rouge, LA
 New research presented at this year’s European Congress on Obesity in Istanbul, Turkey,
shows that use of obesity drugs alongside behavioral interventions leads to a larger
drop in so called ‘food noise’ than in those using the behavioral therapy alone. The
study is by Dr. Hanim Diktas, postdoctoral researcher at LSU’s Pennington Biomedical Research Center in Baton
Rouge, La., and colleagues.
New research presented at this year’s European Congress on Obesity in Istanbul, Turkey,
shows that use of obesity drugs alongside behavioral interventions leads to a larger
drop in so called ‘food noise’ than in those using the behavioral therapy alone. The
study is by Dr. Hanim Diktas, postdoctoral researcher at LSU’s Pennington Biomedical Research Center in Baton
Rouge, La., and colleagues.
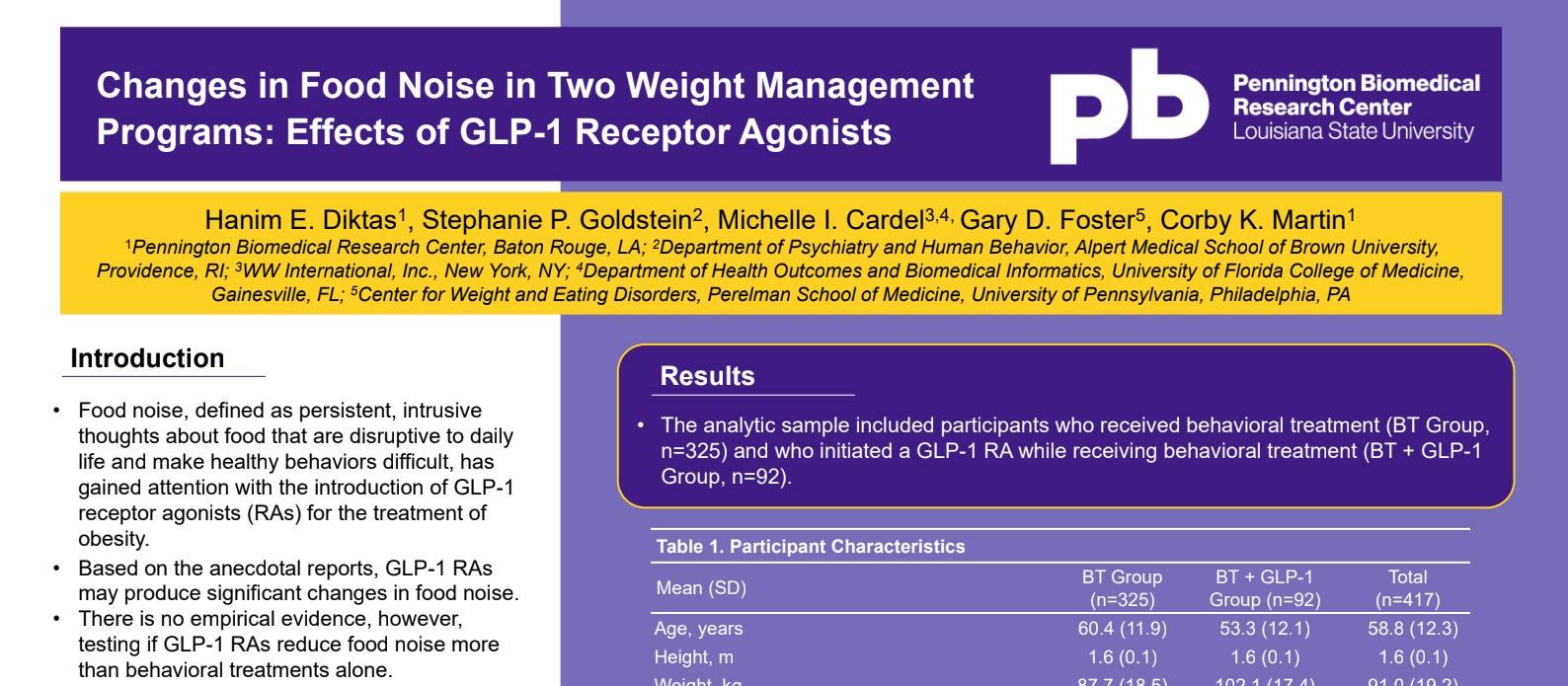
Food noise, defined as persistent, intrusive thoughts about food that are disruptive to daily life and make healthy behaviors difficult, has gained attention with the introduction of GLP-1 receptor agonists (RAs) for the treatment of obesity. Based on anecdotal reports, GLP-1 RAs may produce significant changes in food noise. There is no empirical evidence, however, testing if GLP-1 RAs reduce food noise more than behavioral treatments alone. This study examined short-term changes in food noise in a digital behavioral weight management program with and without GLP-1 RAs.
Dr. Diktas presented “Changes in Food Noise in Two Weight Management Programs: Effects of GLP-1 Receptor Agonists” during the poster sessions of the 33rd European Congress on Obesity held this week.
This observational cohort study included 417 adults participating in a digital behavioral weight management program and evaluated one-month change in food noise using the validated Food Noise Questionnaire* (FNQ) and compared outcomes between those with and without GLP-1 receptor agonist use.
The FNQ asks participants five questions and scores each answer from 0-to-4 points to give a maximum total of 20. The questions/statements that users must rank themselves on, with the ratings strongly disagree; disagree; neither agree nor disagree; agree; strongly agree, are as follows:
- I find myself constantly thinking about food throughout the day.
- My thoughts about food feel uncontrollable.
- I spend too much time thinking about food.
- My thoughts about food have negative effects on me and/or my life.
- My thoughts about food distract me from what I need to do.
All individuals completed an online survey at baseline and a month follow-up, the analytic sample included participants who initiated a GLP-1 RA alongside behavioral treatment (“Weight Watchers Med+, GLP-1 RA,” n=92) and participants who did not initiate a GLP-1 RA while receiving behavioral treatment (“Weight Watchers Core+, No GLP-1 RA,” n=325). Individuals using GLP-1 RAs or any weight loss medications at baseline were not included.
FNQ scores and body weight measured with digital scales were collected at baseline and at the one-month follow-up. Statistical modelling estimated change over time and whether the change differed between the two groups. All statistical models were adjusted for baseline FNQ scores; which allowed us to analytically isolate the effect of GLP-1 use on changes in FNQ scores.
Most participants were white (390, 94%) and female (388, 93%), with a mean age of 59 years and a mean BMI of 34 kg/m2. The FNQ scores at baseline were significantly different between the two groups. In Weight Watchers Med+, mean FNQ (unadjusted) decreased from 13.1 to 8.7; in Weight Watchers Core+, mean FNQ (unadjusted) decreased from 10.7 to 9.7.
Statistical models adjusted for baseline FNQ revealed significant decreases in food noise from baseline to one-month follow-up for the Weight Watchers Med+ (adjusted mean change -4.05), and the Weight Watchers Core+ group (-1.15). The results also indicated a greater decrease in food noise in the Weight Watchers Med+ group (adjusted between-groups difference -3.0).
Additional analyses examining weight change and its relationship to changes in food noise are underway for the full manuscript.
The authors say, “Food noise was reduced during weight loss treatment, and initiation of GLP-1 RA drugs was associated with a significantly greater reduction than behavioral treatment alone. This substantial reduction in food noise with GLP1 RA drugs, when combined with behavioral treatment, is in line with previous anecdotal findings and may serve as an early indicator of treatment response.
“The current study focused on the one‑month follow-up because it was the first follow-up time point available in this dataset for most participants and provided indication of short-term change in food noise. However, future studies should investigate food noise over a longer follow-up period to understand the mechanistic role of food noise in weight, health and quality of life outcomes.”
Notes to editors – link to food noise questionnaire https://onlinelibrary.wiley.com/doi/10.1002/oby.24216.
For more information on the food noise questionnaire, how to use and translations hub, visit www.pbrc.edu/fnq.
Dr. Hanim Diktas, Pennington Biomedical Research Center, hanim.diktas@pbrc.edu
Tony Kirby in the ECO Media Centre T) +44 7834 385827 E) tony.kirby@tonykirby.com
Conflict of interest and funding: Gary D. Foster and Michelle I. Cardel were employees of Weight Watchers (WW) International, Inc., and Michelle I. Cardel is a shareholder of WW. The research that is conducted on the development and validation of the Food Noise Questionnaire (FNQ) was funded by WWInc. via a research contract awarded to Pennington Biomedical Research Center. Research relating to this abstract was funded by WW International, Inc. Pennington Biomedical Research Center is supported by Nutrition Obesity Research Center (NORC) grant P30 DK072476 titled “Nutrition and Metabolic Health Through the Lifespan” sponsored by the National Institute for Diabetes and Digestive and Kidney Diseases (NIDDK) and by grant U54 GM104940 from the National Institute of General Medical Sciences, which funds the Louisiana Clinical and Translational Science Center. Hanim E. Diktas is supported by Postdoctoral Fellowship (F32 HD116537).
This press release is based on poster abstract 1276 at the European Congress on Obesity (ECO) in Istanbul, Turkey. All accepted abstracts have been extensively peer reviewed by the congress selection committee. There is no full paper at this stage, but the authors are happy to answer your questions. The research is in preparation for submission to the Nutrition & Diabetes journal for publication.
For full abstract, click here.
For full poster, click here.
For more information contact:
Ernie Ballard, Senior Director of Communications & Marketing, ernie.ballard@pbrc.edu, 225-263-2677.
About the Pennington Biomedical Research Center
The Pennington Biomedical Research Center is at the forefront of medical discovery as it relates to understanding the triggers of obesity, diabetes, cardiovascular disease, cancer and dementia. Pennington Biomedical has the vision to lead the world in promoting nutrition and metabolic health and eliminating metabolic disease through scientific discoveries that create solutions from cells to society. The Center conducts basic, clinical and population research, and is a campus in the LSU System.
The research enterprise at Pennington Biomedical includes over 600 employees within a network of 44 clinics and research laboratories, and 16 highly specialized core service facilities. Its scientists and physician/scientists are supported by research trainees, lab technicians, nurses, dietitians and other support personnel. Pennington Biomedical is a globally recognized state-of-the-art research institution in Baton Rouge, Louisiana. For more information, see www.pbrc.edu.
Pennington Biomedical Research Center
6400 Perkins Road
Baton Rouge, LA 70808



