News Feed

Pennington Biomedical and the U.S. Military
Since 1988, the Pennington Biomedical Research Center has supported the U.S. Military and has become the No. 1 provider of nutrition science for the Department of Defense, or DOD. Over the years, that relationship has grown to include research focused on all aspects of readiness, performance and resilience. Over the past 35 years, Pennington Biomedical has attracted researchers from around the world to its Baton Rouge campus with the purpose of helping people live longer, healthier lives. These internationally renowned scientists collaborate with their colleagues in 240 academic institutions in 24 countries. Their work has generated close to 400,000 citations in scientific literature with five scientists ranking among the most cited researchers in the world
Learn More
Pennington Biomedical’s Dr. Steven Heymsfield and Colleague Publish Guidance on Energy and Macronutrients Across the Life Span
In the long history of recommendations for nutritional intake, current research is trending toward the concept of “food as medicine” – a philosophy in which food and nutrition are positioned within interventions to support health and wellness. In the paper – “Guidance on Energy and Macronutrients Across the Lifespan” – by Pennington Biomedical Research Center’s Dr. Steven Heymsfield, he shares the latest clarity and recommendations in the rich and storied history of energy and macronutrient intake.
Learn More
Ways to Work Out in the Workplace
Chances are you’ve heard it before: To stay in shape, you need to eat less and move more. Eating less may be difficult. But, moving more may seem nearly impossible given the demands of family, school and work—not to mention the commute among all three. Over the past 35 years, Pennington Biomedical has attracted researchers from around the world to its Baton Rouge campus with the purpose of helping people live longer, healthier lives. These internationally renowned scientists collaborate with their colleagues in 240 academic institutions in 24 countries. Their work has generated close to 400,000 citations in scientific literature with five scientists ranking among the most cited researchers in the world
Learn More
March 2024 Publications
The following are recent publications by the researchers at the Pennington Biomedical Research Center.
Learn More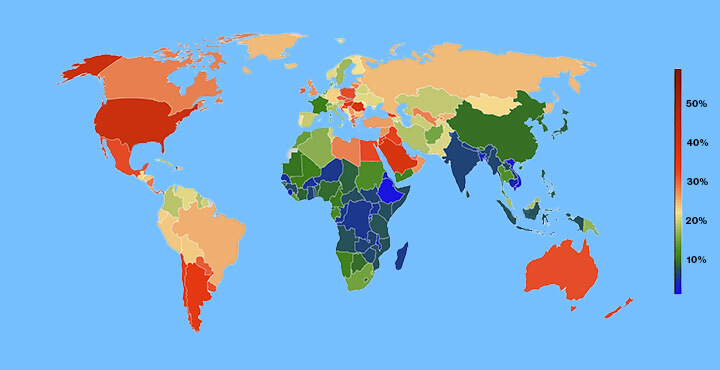
More Than One Billion People Around the Globe are Facing Obesity
Obesity is now the most common form of malnutrition in most countries, with the rates rising in all categories of men, women, children and adolescents since 1990. Obesity rates in adult men have nearly tripled since 1990, with obesity rates in woman doubling in that time. The rates of obesity in children and adolescents quadrupled in that time frame, as well. As of 2022, nearly 880 million adults and nearly 160 million children were living with obesity.
Learn More
Comparative Study of Type 2 Diabetes Medications Shows Differences in Medication Acceptance, Quality-Of-Life, Insulin Secretion and Mortality
Diabetes affects more than 1 in 10 — or more than 38 million — Americans. People with diabetes who keep their blood sugar levels in the near-normal range generally have a much lower risk of developing diabetes complications such as heart, kidney, and eye diseases. The challenge is that most people with diabetes require more than one medication to control blood sugar levels over time.
Learn More
A Father's Legacy: Dr. Justin Brown's Cancer Research
What do you do when one of the world's deadliest diseases hits your home? You use it as fuel. Meet Dr. Justin Brown, Director of Pennington Biomedical's Cancer Metabolism Program, and hear the inspirational story behind his passion. Every 18 seconds someone is diagnosed with cancer in the United States and every 52 seconds someone succumbs to this devastating disease. Individuals living with cancer and those at risk for developing cancer deserve to know how purposeful changes to their lifestyle can alter their clinical course. Over the past 35 years, Pennington Biomedical has attracted researchers from around the world to its Baton Rouge campus with the purpose of helping people live longer, healthier lives. These internationally renowned scientists collaborate with their colleagues in 240 academic institutions in 24 countries. Their work has generated close to 400,000 citations in scientific literature with five scientists ranking among the most cited researchers in the world
Learn More
Health Across the Lifespan Begins with Small Shifts
At Pennington Biomedical, our passion is straightforward: prevent disease, treat disease, end disease. Through innovative research, and with the involvement of the Baton Rouge community, we are applying cutting edge technologies to improve human health. But in addressing nutrition, obesity and diabetes, many approaches do not employ state-of-the-art lab equipment. Addressing them requires small shifts.
Learn More
National Colorectal Cancer Awareness Month: A Father’s Legacy Leads to a Research Passion for Dr. Justin Brown
Growing up, Dr. Justin Brown thought his career path would bring him to be a union carpenter in New York City, but when he was 14 years old, his father passed away following a battle with colon cancer. From then on, Dr. Brown’s future career became instantly clear. “My mission is motivated by the need to prevent children from having to watch their parents melt away from cancer,” Dr. Brown said.
Learn More
Pennington Biomedical Research Center Named a Spoke in the ARPA-H Investor Catalyst Hub
Shortly after being selected as a spoke in the ARPA-H Customer Experience Hub, the Pennington Biomedical Research Center has also been named a spoke in the ARPA-H Investor Catalyst Hub.
Learn More
A Scientific Center Becomes an Economic Engine
Entrepreneurial scientists at LSU’s Pennington Biomedical Research Center win national grants and corporate contracts that fund hundreds of biomed jobs at the Center in Baton Rouge each year. Over the past 35 years, Pennington Biomedical has attracted researchers from around the world to its Baton Rouge campus with the purpose of helping people live longer, healthier lives. These internationally renowned scientists collaborate with their colleagues in 240 academic institutions in 24 countries. Their work has generated close to 400,000 citations in scientific literature with five scientists ranking among the most cited researchers in the world
Learn More
Pennington Biomedical to Host the 3rd Annual Senior Black American Health Fair on Saturday, March 30
Open to residents in the Greater Baton Rouge region and beyond, the health fair will take place on Saturday, March 30, from 8:30 a.m. to 1 p.m. at the C.B. Pennington Conference Center Building, located at 6400 Perkins Road in Baton Rouge.
Learn More
If You Can Only Work Out on the Weekends, Here’s How Long You Need to Exercise
New research suggests that people who cram their sweat sessions into a weekend have body fat levels that resemble those who hit the gym throughout the week, as long as they're hitting at least 150 minutes of exercise total.
Learn More
Inaugural Neuroscience Symposium: Bringing Minds Together
The Baton Rouge Chapter of the Society for Neuroscience Baton Rouge Chapter (SfN BTR) presents the inaugural “Neuroscience Symposium: Bringing Minds Together.” The event will take place Friday, March 8 from 8:30 a.m. to 4:15 p.m. at Pennington Biomedical Research Conference Center, in Baton Rouge, Louisiana.
Learn More
Remote Height and Weight Measurements for Children Proven as Accurate as In-Person
The increased popularity of telehealth and telemedicine in recent years has provided a new level of accessibility for patients to visit doctors and learn more about their health. Certain remote measurements, however, have long been speculated to be less accurate and less trustworthy than when gathered at in-person appointments.
Learn More
February 2024 Publications
The following are recent publications by the researchers at the Pennington Biomedical Research Center.
Learn More
Bariatric Surgery Provides Long-Term Blood Glucose Control, Type 2 Diabetes Remission
People with type 2 diabetes who underwent bariatric surgery achieved much better long-term blood glucose control compared to people who received medical management plus lifestyle interventions, according to a new study published in JAMA, or Journal of the American Medical Association, and funded by the National institute of Diabetes, Digestive and Kidney Diseases, part of The National Institutes of Health.
Learn More
Pennington Biomedical Researchers Looking for New Ways to Improve Life Span and Health During Aging
The Pennington Biomedical Research Center is discovering new ways to improve life span and health during aging through its research. A new study from Pennington Biomedical, “Restricting bioenergetic efficiency enhances longevity and mitochondrial redox capacity in Drosophila melanogaster,” published in the journal Aging Cell, showed that BAM15, a compound that makes mitochondria less efficient at producing energy, extended life span in fruit flies, and was associated with less body fat and increased muscle function.
Learn More
New Screening Tool Saving Lives: Metamor Team Developing Tool to Catch Cancer Earlier
A new partnership between a Pennington Biomedical physician-researcher at the Metamor Institute and an LSU School of Medicine oncologist has already saved several women’s lives in southeast Louisiana.
Learn More
Whether You Exercise Regularly or One-To-Two Days a Week, Weight Loss is Possible
Whether you engage in physical activity on a regular basis or one-to-two days a week, both options produce weight loss suggests a new study published in the journal Obesity, The Obesity Society’s (TOS) flagship journal. The study is the first of its kind to examine the association between physical activity patterns and objectively-measured fat tissue mass.
Learn More


